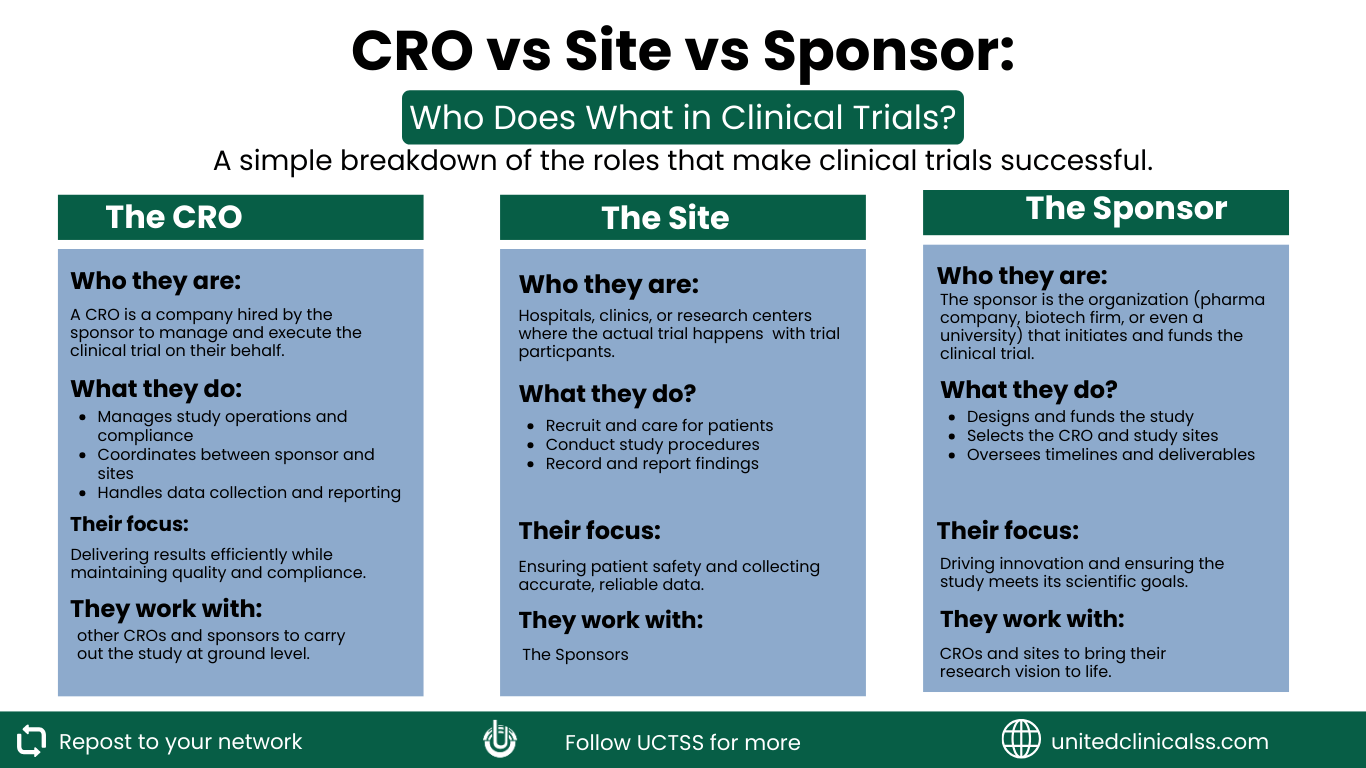
Behind every clinical trial that progresses from protocol approval to patient enrolment, three groups of organisations work in parallel, each responsible for a distinct layer of the research process. These are the sponsor, the contract research organisation (CRO), and the clinical research site.
Each of which plays a key part in ensuring that these studies are conducted safely and effectively, especially for patients considering participation.
As a Clinical research professional, understanding how these interact helps clarify job descriptions, reporting lines, and day-to-day responsibilities and reveals where the different career lathways sit within the industry
Who are the sponsors in clinical trials?:
The sponsor is the organisation that initiates a clinical trial. In most cases, that is a pharmaceutical company, a biotechnology firm, or an academic institution with research funding.
The sponsor owns the investigational product and the intellectual framework behind it. More importantly, the sponsor carries ultimate regulatory and ethical responsibility for the trial, regardless of how much operational work is outsourced.
If data is compromised, a safety signal is missed, or a protocol is not followed, the regulatory consequences fall to the sponsor. This is why sponsor oversight is a structural requirement.
The sponsor remains accountable for every element of the trial, regardless of whether they executed it directly.
What the Sponsor Is Responsible For
• Designing the protocol and defining the scientific objectives of the trial
• Providing the investigational product, funding, and financial support
• Selecting the CRO, clinical sites, and other vendors
• Maintaining regulatory submissions and communicating with regulatory authorities
• Overseeing safety reporting and approving any protocol amendments
• Conducting or commissioning the final data analysis and regulatory dossier
Sponsors range from large multinational pharmaceutical companies with thousands of internal staff to small biotech startups that have never run a trial before.
The CRO:
A contract research organisation is a company engaged by the sponsor to plan and execute some or all of the clinical trial’s operational activities. By acting as intermediaries between sponsors and sites, they help ensure that the trial runs smoothly and according to plan.
A CRO bridges the gap between a sponsor’s scientific vision and its practical execution on the ground. CROs bring specialised teams, established systems, and cross-regional experience that sponsors would otherwise take years to build internally.
What the CRO Is Responsible For
• Managing study start-up activities, including site feasibility assessments and contract negotiations
• Coordinating regulatory submissions in each relevant country or jurisdiction
• Hiring and deploying Clinical Research Associates (CRAs) to monitor sites
• Overseeing data collection, query resolution, and data management
• Supporting safety reporting and communicating medical events to the sponsor
• Training site staff on the protocol and Good Clinical Practice (GCP) requirements
• Maintaining the Trial Master File (TMF) and ensuring audit-readiness
The CRO manages that relationship day to day, ensuring sites are supported, issues are escalated, and reporting is accurate and timely.
The CRO is the operational engine of a clinical trial. It translates the sponsor’s protocol into structured, monitored, compliant activity across every site.
The Clinical Research Site:
The clinical site is where the trial actually happens. These include the hospitals, research clinics, or academic medical centres where patients are enrolled, treated, and followed up according to the protocol. Everything that the sponsor designs and the CRO coordinates ultimately depends on what happens at the site level.
The Principal Investigator (PI) leads each site and holds direct legal and ethical responsibility for the participants enrolled. Supporting the PI is a team that typically includes sub-investigators, clinical research coordinators, study nurses, and data entry staff. Together, this team is the human face of the trial for every patient who walks through the door.
What the Site Is Responsible For
• Identify and screen potential participants in line with the study’s eligibility criteria
• Ensure informed consent is properly obtained and documented for every participant
• Carry out all study visits, assessments, and trial procedures in strict accordance with the approved protocol
• Entering accurate and complete data into the Electronic Data Capture (EDC) system
• Report adverse events and serious adverse events within the required regulatory timelines
• Maintain complete and well-organised source documents and make them available for monitoring and audit
• Responding to queries raised by the CRO or sponsor promptly
The site is also where data quality is made or broken. Source documents at the site, which include patient notes, lab results, imaging reports, and consent forms, are the foundation against which everything entered into the EDC is verified. Errors at source cannot always be corrected downstream, which is why training, documentation discipline, and a responsive site team are so critical to trial quality.
Where the Three Roles Meet
The sponsor relies on the CRO’s reporting to make oversight decisions. The CRO relies on the site’s data to fulfil its monitoring obligations. The site relies on the CRO for training and query resolution, and on the sponsor’s protocol for clinical direction.
Defined roles, clear contracts, and consistent communication between sponsor, CRO, and site are what protect patient safety and data quality across the entire trial.
UCTSS is a Contract Research Organisation working to advance early and late-phase clinical trial research in Nigeria and West Africa.
We work across all three of these relationships. We support sites in building the operational capacity to run trials to international GCP standards and partner with sponsors bringing studies into Nigeria and across West Africa.
As a CRO that understands the regulatory environment, the practical realities of the ecosystem, and the standards that global sponsors expect.

Add a Comment